Clinically Proven Non-Opioid Pain Relief
Cold therapy has been used for decades to support post-operative recovery by reducing localized inflammation and discomfort.
Reduces pain and swelling4,7,8,9,10
Reduces opioid consumption
Speeds up post-operative rehabilitation and recovery.
Effective for a variety of indications and protocols.
Dr. Sarah Edwards discusses the NOPAIN Act.
Cold therapy may reduce pain, narcotic use after arthroscopic rotator cuff repair
Results showed patients who undergo arthroscopic rotator cuff repair may experience a reduction in pain and narcotic use with a postoperative cold therapy regimen.
IceMan® Delivers Consistency You Can Count On
Trusted systems engineered for controlled cold therapy.
Designed for hospital and ASC environments, IceMan® systems deliver controlled cold therapy across a range of surgical applications.
Time-Tested Cold Therapy
IceMan® supports extended cold therapy across a range of surgical indications and post-operative care protocols.
IceMan® Classic3
The IceMan® helps provide extended cold therapy to aid in a variety of indications and protocols as directed by a medical professional.
IceMan® Clear3
The IceMan CLEAR3 cold therapy unit helps reduce pain and swelling, speeding up rehabilitation and recovery.
How IceMan® Delivers
DonJoy’s recirculation system helps maintain more consistent and accurate temperatures than other competitive cold therapy units.
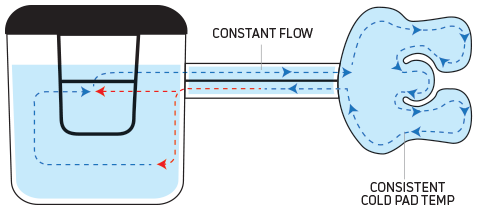
ICEMAN with dual pump recirculation technology. The DonJoy® IceMan® features a recirculation system that allows water warmed after flowing through the cold pad to be preserved and remixed with cooler ice water at a constant flow rate, providing consistent cool water distribution throughout the cold pad.
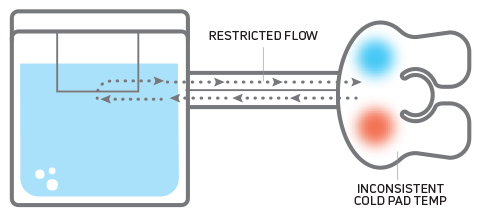
COMPETITORS Other versions restrict or pinch water flow to the cold pad, causing variable flow rates and inconsistent cold pad temperatures.
A Clear Path to Medicare Reimbursement
Under the NOPAIN Act, qualifying non-opioid pain management therapies are eligible for separate Medicare facility payment in hospital outpatient departments and Ambulatory Surgery Centers. Eligible IceMan® systems may be reimbursed when billed in accordance with CMS guidance. Facilities should verify eligibility with their Medicare Administrative Contractor (MAC) prior to submission.
Discuss eligibility, product options, and reimbursement with a sales rep.
Separate Medicare Facility Payment
Qualifying IceMan® devices are eligible for separate Medicare facility payment when billed in accordance with CMS guidelines.
Billing and Claims Submission
Facilities submit claims using the appropriate HCPCS code in accordance with CMS guidance. Reimbursement eligibility should be verified with your MAC prior to submission.
FAQs
Is a DMEPOS license required for a facility to be able to bill and be paid by traditional Medicare for HCPCS code C9810?
No. Hospitals or ASCs can apply the DonJoy® IceMan® to the patient as part of the surgical procedure. Traditional Medicare claims will be submitted by the hospital or ASC to the Medicare Part B Medicare Administrative Contractor (MAC), and payments will be made through the Medicare Hospital Outpatient Prospective Payment System (OPPS) or through the Ambulatory Surgery Center Payment System, rather than through the DMEPOS MACs.
Define what is a C-Code?
A C-code is a CMS-established billing code that is reportable by certain outpatient facilities (hospitals, ASCs) in connection with items and services provided in those settings. C-codes have historically been used to report transitional pass-through drugs and devices; new procedures recognized by CMS under the New Technology APC pathway; and now, NOPAIN Act eligible drugs and devices. They are generally not reportable by other types of providers (physicians, DME suppliers, etc.).
Is the C9810 code specific to the NOPAIN Act? Does E0218 remain the correct HCPCS code to report for all non-eligible NOPAIN Act billing scenarios?
Yes, HCPCS code C9810 is specific to use for the DonJoy® IceMan® device in connection with a traditional Medicare covered procedure performed in an HOPD or ASC, when purchased by and dispensed by the hospital or ASC performing the procedure.
Other codes may apply for DonJoy® IceMan® use in inpatient or home settings (e.g., code E0218) outside of the hospital outpatient or ASC settings.
Can the product be brought and used at home?
The product should be dispensed and applied by the hospital or ASC prior to discharge. The patient can take the IceMan® home, however, to continue to manage post-surgical pain.
Does the traditional Medicare payment go through the DMEPOS MAC route or through the traditional Medicare Part B facility payment route?
The traditional Medicare facility payment route. The add-on payment available under the NOPAIN Act will be made to the hospital or ASC by the Part B MAC, NOT by the DME MAC.
Is there a list of approved procedures that qualify for the NOPAIN Act? Can you provide guidance on the types of procedures that qualify?
No, there are no approved or “covered procedure” lists under the NOPAIN Act associated with specific drugs or devices. Under the NOPAIN Act, CMS designates qualifying non opioid drugs, biologics, and devices for separate payment when they are furnished in connection with any covered outpatient surgical service in hospital outpatient departments (HOPDs) or ambulatory surgery centers (ASCs), subject to standard traditional Medicare coverage limitations (i.e., the item must be “reasonable and necessary” for the treatment of the patient). CMS does not prevent facilities from billing for qualifying non-opioid products alongside any covered outpatient surgical procedure.
Will the separate payment lead to a reduction in compensation for the facilities and/or providers for the procedures they are also billing?
Not at this time. The NOPAIN Act provides an add-on payment by traditional Medicare Part B to the hospitals or ASCs that use eligible non-opioid drugs or devices, separate and apart from the procedure reimbursement.
What qualifies as an outpatient procedure? Please confirm, single overnight, less than 23 hours.
The procedure is performed in a hospital outpatient setting or an ASC setting, and the patient is not later admitted to a hospital for that procedure in the following 72 hours. The “two midnight” rule is still in effect. This rule applies to situations in which CMS generally believes a hospital encounter should be considered an “inpatient” admission (rather than “outpatient” procedure) if the physician reasonably expects the patient will be in the hospital for two or more midnights.
If a customer asked how much to bill, what is our reply?
Enovis cannot advise providers on the amount to be charged or billed. Providers should work with their Revenue Cycle Management (RCM) and Chargemaster teams to determine an appropriate amount to bill traditional Medicare, consistent with their current policies on establishing charges and billed amounts.
How is the product monitored by traditional Medicare/CMS to ensure that it is a qualified device (e.g., DonJoy® IceMan®)?
The HCPCS code established for the DonJoy® IceMan® (C9810) is specific to the DonJoy® IceMan® device. Providers may be subject to audits and claims reviews by CMS and by the CMS Part B MACs to ensure compliance with coding and billing requirements, including to ensure that only qualified devices are used/reported with HCPCS code C9810.
Please confirm this is not exclusive to orthopedics, musculoskeletal procedures. For example, plastic surgery would also be eligible.
There are no traditional Medicare approved or “covered procedure” lists or specialties associated with specific drugs or devices under the NOPAIN Act. However, be aware that traditional Medicare does NOT typically cover/pay for plastic/cosmetic surgery procedures.
Is the traditional Medicare reimbursement for HCPCS code C9810 subject to Medicare’s 80/20 DME coverage? Or will the patient not have a 20% coinsurance amount since this is a non-DME facility reimbursement?
As an item covered under traditional Medicare Part B, reimbursement for HCPCS code C9810 to a hospital outpatient department or to an ASC would be subject to the patient’s 20% coinsurance amount.
Do providers have experience billing in this manner? Or is this a new billing method?
Many hospitals and ASC providers have experience reporting C-codes for pass-through medical devices or for new procedures
Additional Resources
Overview & FAQs
Final NOPAIN Ruling
Billing and Reimbursement Guides
- Detailed Billing Guide
- DonJoy® Iceman® CMS 1450 Example
- DonJoy® Iceman® CMS 1500 Example
- Breakdown of Outpatient Prospective Payment System (OPPS) Final Rule
Medicare Program Final Ruling
Under the federal NOPAIN Act, effective Jan. 1, 2026, qualifying IceMan® systems are eligible for separate Medicare facility payment in HOPDs and ASCs.
- Xiao AX, et al. The use of a motorized cold therapy unit in the postoperative period influences pain and narcotic use following arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2025; [Epub ahead of print]. doi:10.1016/j.jse.2025.08.026.
- Shah A, Hayes CJ, Martin BC. Characteristics of initial prescription episodes and likelihood of long-term opioid use—United States, 2006–2015. MMWR Morb Mortal Wkly Rep. 2017;66(10):265–269.
- Bicket MC, Long JJ, Pronovost PJ, et al. Prescription opioid analgesics commonly unused after surgery: a systematic review. JAMA Surg. 2017;152(11):1066–1071.
- Raynor MC, et al. Cryotherapy after ACL reconstruction. J Knee Surg. 2005;18(3):123–129.
- Shelbourne KD, et al. Postoperative cryotherapy for the knee in ACL reconstructive surgery. Orthopedics International Edition. 1994.
- Jones CA, et al. Opioid-sparing pain management protocol after shoulder arthroplasty results in less opioid consumption and higher satisfaction: a prospective, randomized controlled trial. J Shoulder Elbow Surg. 2002;11(2):20–25.
- Levy AS, Marmar M. The role of cold compression dressings in the postoperative treatment of total knee arthroscopy. Clin Orthop Relat Res. 1993;297:174–178.
- Barber FA. A comparison of crushed ice and continuous flow cold therapy. Am J Knee Surg. 2000;13(1):1–8.
- Mumith A, et al. Enhancing postoperative rehabilitation following knee arthroplasty using a new cryotherapy product: a prospective study. Geriatr Orthop Surg Rehabil. 2015;6(2):129–135.
- Singh H, et al. The efficacy of continuous cryotherapy on the postoperative shoulder: a prospective, randomized investigation. Orthop Clin North Am. 2001;32(3):491–497.
- Su EP, et al. A prospective, multicenter, randomized trial to evaluate the efficacy of a cryopneumatic device on total knee arthroplasty recovery. J Bone Joint Surg Am. 2012;94(18):1531–1537.
- Martimbianco AL, Gomes da Silva BN, de Carvalho AP, Silva V, Torloni MR, Peccin MS. Effectiveness and safety of cryotherapy after arthroscopic anterior cruciate ligament reconstruction: a systematic review of the literature. Phys Ther Sport. 2014;15(4):261–268.
- Department of Health and Human Services, Centers for Medicare & Medicaid Services. Medicare Program: Hospital Outpatient Prospective Payment and Ambulatory Surgical Center Payment Systems; Quality Reporting Programs; Overall Hospital Quality Star Rating; Hospital Price Transparency; and Notice of Closure of a Teaching Hospital and Opportunity To Apply for Available Slots. Federal Register, 25 Nov. 2025, https://public-inspection.federalregister.gov/2025-20907.pdf

